How To Draw The Probability Function In Quantum In 2d
Quantum Mechanics
53 The Quantum Particle in a Box
Learning Objectives
By the stop of this section, you will be able to:
- Describe how to prepare a boundary-value trouble for the stationary Schrӧdinger equation
- Explain why the energy of a quantum particle in a box is quantized
- Describe the physical meaning of stationary solutions to Schrӧdinger'southward equation and the connection of these solutions with time-dependent quantum states
- Explain the physical meaning of Bohr'south correspondence principle
In this section, we apply Schrӧdinger'south equation to a particle bound to a 1-dimensional box. This special example provides lessons for understanding quantum mechanics in more circuitous systems. The energy of the particle is quantized as a consequence of a standing wave condition within the box.
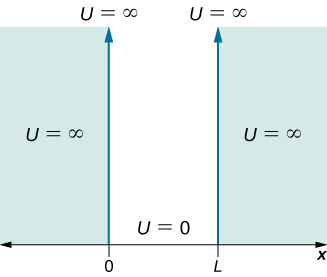
Consider a particle of mass ![]() that is immune to move only along the x-direction and its motion is bars to the region between hard and rigid walls located at
that is immune to move only along the x-direction and its motion is bars to the region between hard and rigid walls located at ![]() and at
and at ![]() ((Figure)). Between the walls, the particle moves freely. This physical situation is called the infinite square well, described by the potential energy role
((Figure)). Between the walls, the particle moves freely. This physical situation is called the infinite square well, described by the potential energy role
![]()
Combining this equation with Schrӧdinger's time-independent wave equation gives
![]()
where Due east is the full free energy of the particle. What types of solutions exercise we expect? The energy of the particle is a positive number, then if the value of the wave part is positive (right side of the equation), the curvature of the wave role is negative, or concave downwards (left side of the equation). Similarly, if the value of the wave function is negative (right side of the equation), the curvature of the wave function is positive or concave upwards (left side of equation). This condition is met by an aquiver wave role, such as a sine or cosine wave. Since these waves are confined to the box, we envision standing waves with stock-still endpoints at ![]() and
and ![]() .
.
The potential energy function that confines the particle in a 1-dimensional box.
Solutions ![]() to this equation accept a probabilistic interpretation. In item, the square
to this equation accept a probabilistic interpretation. In item, the square ![]() represents the probability density of finding the particle at a particular location ten. This function must be integrated to make up one's mind the probability of finding the particle in some interval of infinite. We are therefore looking for a normalizable solution that satisfies the following normalization condition:
represents the probability density of finding the particle at a particular location ten. This function must be integrated to make up one's mind the probability of finding the particle in some interval of infinite. We are therefore looking for a normalizable solution that satisfies the following normalization condition:
![]()
The walls are rigid and impenetrable, which means that the particle is never found beyond the wall. Mathematically, this means that the solution must vanish at the walls:
![]()
Nosotros wait oscillating solutions, so the most general solution to this equation is
![]()
where yard is the moving ridge number, and ![]() and
and ![]() are constants. Applying the boundary status expressed by (Figure) gives
are constants. Applying the boundary status expressed by (Figure) gives
![]()
Because we have ![]() , the solution must exist
, the solution must exist
![]()
If ![]() is zero,
is zero, ![]() for all values of 10 and the normalization condition, (Effigy), cannot be satisfied. Bold
for all values of 10 and the normalization condition, (Effigy), cannot be satisfied. Bold ![]() , (Effigy) for
, (Effigy) for ![]() then gives
then gives
![]()
Nosotros discard the ![]() solution because
solution because ![]() for this quantum number would exist cipher everywhere—an un-normalizable and therefore unphysical solution. Substituting (Figure) into (Figure) gives
for this quantum number would exist cipher everywhere—an un-normalizable and therefore unphysical solution. Substituting (Figure) into (Figure) gives
![]()
Calculating these derivatives leads to
![]()
According to de Broglie, ![]() and so this expression implies that the full energy is equal to the kinetic energy, consistent with our assumption that the "particle moves freely." Combining the results of (Figure) and (Figure) gives
and so this expression implies that the full energy is equal to the kinetic energy, consistent with our assumption that the "particle moves freely." Combining the results of (Figure) and (Figure) gives
![]()
Strange! A particle bound to a one-dimensional box can only have certain discrete (quantized) values of energy. Farther, the particle cannot accept a cipher kinetic energy—it is incommunicable for a particle bound to a box to be "at residue."
To evaluate the allowed moving ridge functions that stand for to these energies, nosotros must find the normalization abiding ![]() . We impose the normalization condition (Effigy) on the wave role
. We impose the normalization condition (Effigy) on the wave role
![]()
![]()
Hence, the wave functions that correspond to the energy values given in (Figure) are
![]()
For the lowest energy state or footing land energy, nosotros take
![]()
All other energy states tin be expressed as
![]()
The alphabetize n is called the energy quantum number or principal quantum number. The state for ![]() is the kickoff excited state, the land for
is the kickoff excited state, the land for ![]() is the 2nd excited country, and so on. The first iii breakthrough states (for
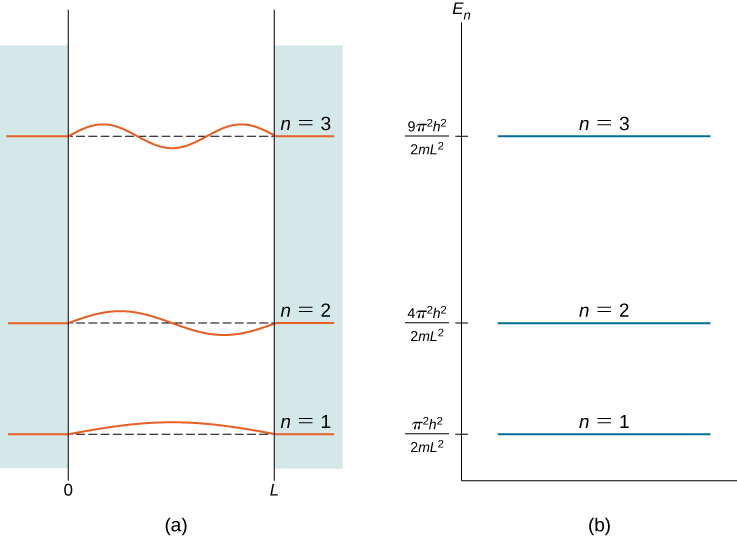
is the 2nd excited country, and so on. The first iii breakthrough states (for ![]() of a particle in a box are shown in (Figure).
of a particle in a box are shown in (Figure).
The wave functions in (Figure) are sometimes referred to as the "states of definite energy." Particles in these states are said to occupy energy levels, which are represented by the horizontal lines in (Figure). Energy levels are coordinating to rungs of a ladder that the particle can "climb" as it gains or loses free energy.
The wave functions in (Effigy) are also chosen stationary country s and standing wave state s. These functions are "stationary," considering their probability density functions, ![]() , do not vary in time, and "standing waves" considering their real and imaginary parts oscillate upward and downwards like a standing wave—like a rope waving between two children on a playground. Stationary states are states of definite energy [(Figure)], but linear combinations of these states, such as
, do not vary in time, and "standing waves" considering their real and imaginary parts oscillate upward and downwards like a standing wave—like a rope waving between two children on a playground. Stationary states are states of definite energy [(Figure)], but linear combinations of these states, such as ![]() (too solutions to Schrӧdinger's equation) are states of mixed free energy.
(too solutions to Schrӧdinger's equation) are states of mixed free energy.
The showtime three breakthrough states of a quantum particle in a box for principal quantum numbers ![]() : (a) standing wave solutions and (b) allowed energy states.
: (a) standing wave solutions and (b) allowed energy states.
Energy quantization is a consequence of the purlieus conditions. If the particle is not bars to a box only wanders freely, the immune energies are continuous. However, in this instance, only certain energies ![]() …) are allowed. The energy divergence betwixt adjacent energy levels is given by
…) are allowed. The energy divergence betwixt adjacent energy levels is given by
![]()
Conservation of free energy demands that if the energy of the arrangement changes, the energy difference is carried in another form of free energy. For the special case of a charged particle bars to a small volume (for example, in an cantlet), free energy changes are oft carried abroad by photons. The frequencies of the emitted photons give us information about the energy differences (spacings) of the system and the book of containment—the size of the "box" [run across (Figure)].
A Simple Model of the Nucleus Suppose a proton is confined to a box of width ![]() (a typical nuclear radius). What are the energies of the footing and the showtime excited states? If the proton makes a transition from the commencement excited land to the footing state, what are the free energy and the frequency of the emitted photon?
(a typical nuclear radius). What are the energies of the footing and the showtime excited states? If the proton makes a transition from the commencement excited land to the footing state, what are the free energy and the frequency of the emitted photon?
Strategy If we presume that the proton confined in the nucleus can exist modeled as a quantum particle in a box, all nosotros need to do is to utilize (Figure) to find its energies ![]() and
and ![]() . The mass of a proton is
. The mass of a proton is ![]() The emitted photon carries away the energy difference
The emitted photon carries away the energy difference ![]() We tin can use the relation
We tin can use the relation ![]() to observe its frequency f.
to observe its frequency f.
Solution The basis state:
![]()
The showtime excited state: ![]() .
.
The energy of the emitted photon is ![]() .
.
The frequency of the emitted photon is
![]()
Significance This is the typical frequency of a gamma ray emitted by a nucleus. The free energy of this photon is about 10 million times greater than that of a visible lite photon.
The expectation value of the position for a particle in a box is given by
![]()
Nosotros can also detect the expectation value of the momentum or average momentum of a large number of particles in a given country:
![Rendered by QuickLaTeX.com \begin{array}{cc}\hfill 〈p〉& =\underset{0}{\overset{L}{\int }}dx{\psi }_{n}^{*}\left(x\right)\left[\text{−}i\hslash \frac{d}{dx}{\psi }_{n}\left(x\right)\right]\hfill \\ & =\text{−}i\hslash \underset{0}{\overset{L}{\int }}dx\sqrt{\frac{2}{L}}\phantom{\rule{0.2em}{0ex}}\text{sin}\phantom{\rule{0.2em}{0ex}}\frac{n\pi x}{L}\left[\frac{d}{dx}\sqrt{\frac{2}{L}}\phantom{\rule{0.2em}{0ex}}\text{sin}\phantom{\rule{0.2em}{0ex}}\frac{n\pi x}{L}\right]=\text{−}i\frac{2\hslash }{L}\underset{0}{\overset{L}{\int }}dx\phantom{\rule{0.2em}{0ex}}\text{sin}\phantom{\rule{0.2em}{0ex}}\frac{n\pi x}{L}\left[\frac{n\pi }{L}\phantom{\rule{0.2em}{0ex}}\text{cos}\phantom{\rule{0.2em}{0ex}}\frac{n\pi x}{L}\right]\hfill \\ & =\text{−}i\frac{2n\pi \hslash }{{L}^{2}}\underset{0}{\overset{L}{\int }}dx\frac{1}{2}\phantom{\rule{0.2em}{0ex}}\text{sin}\phantom{\rule{0.2em}{0ex}}\frac{2n\pi x}{L}=\text{−}i\frac{n\pi \hslash }{{L}^{2}}\phantom{\rule{0.2em}{0ex}}\frac{L}{2n\pi }\underset{0}{\overset{2\text{π}n}{\int }}d\phi \phantom{\rule{0.2em}{0ex}}\text{sin}\phantom{\rule{0.2em}{0ex}}\phi =\text{−}i\frac{\hslash }{2L}·0=0.\hfill \end{array}](https://opentextbc.ca/universityphysicsv3openstax/wp-content/ql-cache/quicklatex.com-95dbeb92094eaf43467d31900c9a7e87_l3.png)
Thus, for a particle in a state of definite energy, the average position is in the centre of the box and the average momentum of the particle is zip—as it would also be for a classical particle. Note that while the minimum energy of a classical particle can be zero (the particle can be at rest in the middle of the box), the minimum energy of a breakthrough particle is nonzero and given past (Figure). The average particle energy in the nth breakthrough state—its expectation value of energy—is
![]()
The outcome is not surprising because the standing wave state is a state of definite energy. Any energy measurement of this organization must return a value equal to one of these allowed energies.
Our analysis of the quantum particle in a box would non be consummate without discussing Bohr's correspondence principle. This principle states that for large breakthrough numbers, the laws of quantum physics must requite identical results as the laws of classical physics. To illustrate how this principle works for a quantum particle in a box, we plot the probability density distribution
![]()
for finding the particle around location x between the walls when the particle is in breakthrough land ![]() . (Figure) shows these probability distributions for the ground state, for the showtime excited state, and for a highly excited state that corresponds to a large quantum number. Nosotros come across from these plots that when a quantum particle is in the ground country, it is most probable to be plant around the middle of the box, where the probability distribution has the largest value. This is not and then when the particle is in the starting time excited state because now the probability distribution has the zero value in the middle of the box, so at that place is no adventure of finding the particle at that place. When a quantum particle is in the first excited state, the probability distribution has ii maxima, and the all-time chance of finding the particle is at positions close to the locations of these maxima. This breakthrough picture is unlike the classical picture.
. (Figure) shows these probability distributions for the ground state, for the showtime excited state, and for a highly excited state that corresponds to a large quantum number. Nosotros come across from these plots that when a quantum particle is in the ground country, it is most probable to be plant around the middle of the box, where the probability distribution has the largest value. This is not and then when the particle is in the starting time excited state because now the probability distribution has the zero value in the middle of the box, so at that place is no adventure of finding the particle at that place. When a quantum particle is in the first excited state, the probability distribution has ii maxima, and the all-time chance of finding the particle is at positions close to the locations of these maxima. This breakthrough picture is unlike the classical picture.
The probability density of finding a classical particle between x and ![]() depends on how much time
depends on how much time ![]() the particle spends in this region. Assuming that its speed u is constant, this time is
the particle spends in this region. Assuming that its speed u is constant, this time is ![]() which is as well constant for whatsoever location between the walls. Therefore, the probability density of finding the classical particle at ten is uniform throughout the box, and in that location is no preferable location for finding a classical particle. This classical flick is matched in the limit of large breakthrough numbers. For instance, when a quantum particle is in a highly excited state, shown in (Figure), the probability density is characterized by rapid fluctuations and so the probability of finding the quantum particle in the interval
which is as well constant for whatsoever location between the walls. Therefore, the probability density of finding the classical particle at ten is uniform throughout the box, and in that location is no preferable location for finding a classical particle. This classical flick is matched in the limit of large breakthrough numbers. For instance, when a quantum particle is in a highly excited state, shown in (Figure), the probability density is characterized by rapid fluctuations and so the probability of finding the quantum particle in the interval ![]() does non depend on where this interval is located between the walls.
does non depend on where this interval is located between the walls.
A Classical Particle in a Box A modest 0.forty-kg cart is moving dorsum and forth along an air track between ii bumpers located 2.0 thou apart. We assume no friction; collisions with the bumpers are perfectly elastic so that between the bumpers, the automobile maintains a constant speed of 0.l m/s. Treating the cart as a quantum particle, estimate the value of the principal quantum number that corresponds to its classical energy.
Strategy We find the kinetic free energy K of the cart and its ground state energy ![]() as though it were a quantum particle. The energy of the cart is completely kinetic, then
as though it were a quantum particle. The energy of the cart is completely kinetic, then ![]() ((Figure)). Solving for north gives
((Figure)). Solving for north gives ![]() .
.
Solution The kinetic energy of the cart is
![]()
The basis country of the cart, treated as a quantum particle, is
![]()
Therefore, ![]() .
.
Significance Nosotros run into from this example that the energy of a classical system is characterized by a very big quantum number. Bohr's correspondence principle concerns this kind of state of affairs. Nosotros can utilise the formalism of quantum mechanics to any kind of arrangement, quantum or classical, and the results are correct in each case. In the limit of high quantum numbers, there is no advantage in using breakthrough formalism because we can obtain the aforementioned results with the less complicated formalism of classical mechanics. However, we cannot apply classical formalism to a quantum system in a low-number free energy state.
Check Your Understanding (a) Consider an infinite square well with wall boundaries ![]() and
and ![]() . What is the probability of finding a quantum particle in its ground state somewhere between
. What is the probability of finding a quantum particle in its ground state somewhere between ![]() and
and ![]() ? (b) Echo question (a) for a classical particle.
? (b) Echo question (a) for a classical particle.
a. nine.i%; b. 25%
Having establish the stationary states ![]() and the energies
and the energies ![]() by solving the time-independent Schrӧdinger equation (Figure), we use (Figure) to write wave functions
by solving the time-independent Schrӧdinger equation (Figure), we use (Figure) to write wave functions ![]() that are solutions of the time-dependent Schrӧdinger's equation given by (Figure). For a particle in a box this gives
that are solutions of the time-dependent Schrӧdinger's equation given by (Figure). For a particle in a box this gives
![]()
where the energies are given past (Effigy).
The quantum particle in a box model has applied applications in a relatively newly emerged field of optoelectronics, which deals with devices that convert electrical signals into optical signals. This model besides deals with nanoscale physical phenomena, such as a nanoparticle trapped in a depression electric potential bounded by loftier-potential barriers.
Summary
- Energy states of a quantum particle in a box are found by solving the time-independent Schrӧdinger equation.
- To solve the time-independent Schrӧdinger equation for a particle in a box and discover the stationary states and allowed energies, we crave that the moving ridge function terminate at the box wall.
- Free energy states of a particle in a box are quantized and indexed by principal quantum number.
- The quantum picture differs significantly from the classical motion picture when a particle is in a depression-free energy state of a low quantum number.
- In the limit of high quantum numbers, when the quantum particle is in a highly excited land, the quantum description of a particle in a box coincides with the classical description, in the spirit of Bohr's correspondence principle.
Conceptual Questions
Using the breakthrough particle in a box model, describe how the possible energies of the particle are related to the size of the box.
Is information technology possible that when we measure the free energy of a quantum particle in a box, the measurement may return a smaller value than the ground land energy? What is the highest value of the energy that nosotros can measure for this particle?
No. For an infinite square well, the spacing between free energy levels increases with the breakthrough number north. The smallest energy measured corresponds to the transition from n = 2 to ane, which is three times the ground state energy. The largest free energy measured corresponds to a transition from ![]() to ane, which is infinity. (Notation: Fifty-fifty particles with extremely large energies remain bound to an infinite foursquare well—they tin never "escape")
to ane, which is infinity. (Notation: Fifty-fifty particles with extremely large energies remain bound to an infinite foursquare well—they tin never "escape")
For a quantum particle in a box, the first excited state ![]() has zip value at the midpoint position in the box, so that the probability density of finding a particle at this point is exactly zero. Explain what is incorrect with the following reasoning: "If the probability of finding a breakthrough particle at the midpoint is zero, the particle is never at this point, right? How does information technology come then that the particle can cantankerous this point on its way from the left side to the right side of the box?
has zip value at the midpoint position in the box, so that the probability density of finding a particle at this point is exactly zero. Explain what is incorrect with the following reasoning: "If the probability of finding a breakthrough particle at the midpoint is zero, the particle is never at this point, right? How does information technology come then that the particle can cantankerous this point on its way from the left side to the right side of the box?
Issues
Assume that an electron in an cantlet can be treated as if information technology were confined to a box of width ![]() . What is the footing state energy of the electron? Compare your result to the ground state kinetic energy of the hydrogen cantlet in the Bohr's model of the hydrogen atom.
. What is the footing state energy of the electron? Compare your result to the ground state kinetic energy of the hydrogen cantlet in the Bohr's model of the hydrogen atom.
9.iv eV, 64%
An electron confined to a box has the ground state energy of 2.five eV. What is the width of the box?
0.38 nm
What is the footing state free energy (in eV) of a proton confined to a one-dimensional box the size of the uranium nucleus that has a radius of approximately xv.0 fm?
What is the footing state energy (in eV) of an ![]() -particle confined to a one-dimensional box the size of the uranium nucleus that has a radius of approximately 15.0 fm?
-particle confined to a one-dimensional box the size of the uranium nucleus that has a radius of approximately 15.0 fm?
1.82 MeV
To excite an electron in a one-dimensional box from its showtime excited state to its third excited land requires 20.0 eV. What is the width of the box?
An electron bars to a box of width 0.xv nm by space potential energy barriers emits a photon when it makes a transition from the first excited state to the basis state. Discover the wavelength of the emitted photon.
24.vii nm
If the energy of the first excited state of the electron in the box is 25.0 eV, what is the width of the box?
Suppose an electron confined to a box emits photons. The longest wavelength that is registered is 500.0 nm. What is the width of the box?
![]()
Hydrogen ![]() molecules are kept at 300.0 Thou in a cubical container with a side length of 20.0 cm. Assume that you can treat the molecules as though they were moving in a 1-dimensional box. (a) Find the ground state energy of the hydrogen molecule in the container. (b) Assume that the molecule has a thermal energy given by
molecules are kept at 300.0 Thou in a cubical container with a side length of 20.0 cm. Assume that you can treat the molecules as though they were moving in a 1-dimensional box. (a) Find the ground state energy of the hydrogen molecule in the container. (b) Assume that the molecule has a thermal energy given by ![]() and find the corresponding quantum number n of the breakthrough land that would correspond to this thermal energy.
and find the corresponding quantum number n of the breakthrough land that would correspond to this thermal energy.
An electron is bars to a box of width 0.25 nm. (a) Draw an energy-level diagram representing the first five states of the electron. (b) Summate the wavelengths of the emitted photons when the electron makes transitions between the fourth and the 2nd excited states, between the 2d excited land and the ground state, and between the third and the second excited states.
a.
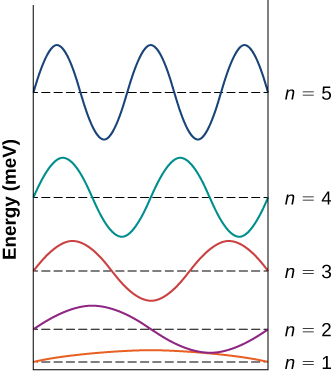
;
b. ![]()
An electron in a box is in the ground country with energy 2.0 eV. (a) Detect the width of the box. (b) How much free energy is needed to excite the electron to its first excited state? (c) If the electron makes a transition from an excited state to the ground state with the simultaneous emission of 30.0-eV photon, find the quantum number of the excited country?
How To Draw The Probability Function In Quantum In 2d,
Source: https://opentextbc.ca/universityphysicsv3openstax/chapter/the-quantum-particle-in-a-box/
Posted by: spiegelanfood.blogspot.com


0 Response to "How To Draw The Probability Function In Quantum In 2d"
Post a Comment